Our endeavour is to help you unlock the full potential of your proteomics research. We do this by providing simple and cost-efficient kits to help you eliminate the most common proteins so that you can enhance the less abundant ones. Looking ahead, we aim to translate this technology into innovative diagnostic tests for common yet underserved diseases, enabling more accurate, personalized treatments and ultimately improving human health.

Mass spectrometry offers exceptional sensitivity for proteomics research; however, its reliability is fundamentally dependent on the quality of sample preparation. Each stage from initial sample collection to the generation of peptide mixtures can introduce variability that impacts data quality and reproducibility (Ignjatovic et al., 2019). To mitigate these challenges, rigorous standards for sample collection and handling have been established. In addition, ready-to-use kits are now available for critical steps such as denaturation, reduction, alkylation, digestion, and peptide purification, ensuring consistent and dependable results throughout your workflow. The same principles apply to the depletion of abundant proteins. Rather than relying on labor-intensive manual protocols, pre-assembled, single-use depletion kits are increasingly favoured by researchers for their ability to:
Mass spectrometry offers exceptional sensitivity for proteomics research; however, its reliability is fundamentally dependent on the quality of sample preparation. Each stage from initial sample collection to the generation of peptide mixtures can introduce variability that impacts data quality and reproducibility (Ignjatovic et al., 2019). To mitigate these challenges, rigorous standards for sample collection and handling have been established. In addition, ready-to-use kits are now available for critical steps such as denaturation, reduction, alkylation, digestion, and peptide purification, ensuring consistent and dependable results throughout your workflow. The same principles apply to the depletion of abundant proteins. Rather than relying on labor-intensive manual protocols, pre-assembled, single-use depletion kits are increasingly favoured by researchers for their ability to:




At Amiprox, our solutions are engineered to deliver consistency, simplicity, and high performance at every stage of your proteomics workflow, empowering you to achieve reliable and reproducible results with confidence.
Mass spectrometry offers exceptional sensitivity for proteomics research; however, its reliability is fundamentally dependent on the quality of sample preparation. Each stage from initial sample collection to the generation of peptide mixtures can introduce variability that impacts data quality and reproducibility (Ignjatovic et al., 2019). To mitigate these challenges, rigorous standards for sample collection and handling have been established. In addition, ready-to-use kits are now available for critical steps such as denaturation, reduction, alkylation, digestion, and peptide purification, ensuring consistent and dependable results throughout your workflow. The same principles apply to the depletion of abundant proteins. Rather than relying on labor-intensive manual protocols, pre-assembled, single-use depletion kits are increasingly favoured by researchers for their ability to:





At Amiprox, our solutions are engineered to deliver consistency, simplicity, and high performance at every stage of your proteomics workflow, empowering you to achieve reliable and reproducible results with confidence.
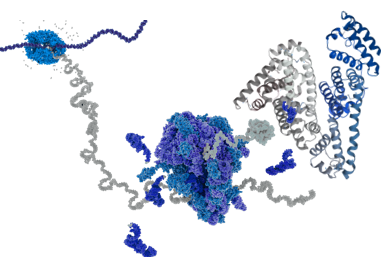
Proteins are large, complex molecules made up of long chains of amino acids. They are essential to all living organisms and perform a vast array of functions within cells and therefore considered to be the building blocks of life. Each protein’s unique structure and function are determined by the specific sequence of its amino acids, which is encoded by the organism’s genetic material (DNA).
Proteins are essential to all living organisms, carrying out a wide range of biological functions. Made of 22 proteinogenic amino acids, their unique sequences, encoded in the genome, define their structure and role. Each organism has its own distinct protein profile, shaped by genetics and environment. The Human Genome Project (2003) paved the way for a new era: proteomics, the systematic study of proteins based on genomic information.
Of the ~19,700 proteins encoded in the human genome, about 93% have been identified (as of 01/2026). Yet, many remain undiscovered, holding the key to unanswered biological and medical questions.
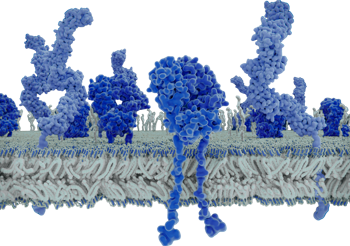
In 1994, Australian scientist Marc Wilkins coined the term proteome, a blend of protein and genome, to describe the complete set of proteins in a cell, tissue, or organism at a given time and condition. Unlike the static genome, the proteome is dynamic, reflecting real-time biological activity through protein types, quantities, and modifications. The large-scale analysis of proteomes, proteomics, only became possible through breakthrough technologies like 2D gel electrophoresis and mass spectrometry, enabling researchers to identify, quantify, and characterize proteins systematically. These tools laid the foundation for modern proteomic research, opening the door to deeper understanding of biology, disease, and potential biomarkers.
Proteomics explore which proteins are produced in the body, where and how they function, and how they interact. More than a standalone field, proteomics is a vital part of genomics broader “omics” sciences like transcriptomics, lipidomics, and metabolomics, together unlocking a holistic understanding of biological systems.
More than a standalone field, proteomics is a vital part of broader “omics” sciences like transcriptomics, lipidomics, and metabolomics, together unlocking a holistic understanding of biological systems.
Proteomics is more than just a complement to genomics. It is essential for decoding life’s functions. By directly studying protein dynamics, it provides unparalleled insight into health and disease.
Proteomics is undergoing a revolution. What once required years of work can now be done in hours: today's advanced mass spectrometry technologies allow us to analyze thousands of proteins at once with remarkable speed, precision, and from the smallest sample volumes. New technologies such as DIA (Data-Independent Acquisition) or single-cell proteomics even make it possible to record the activity of individual cells in detail. Powerful AI-driven analytics turn vast datasets into meaningful patterns that help distinguish health from disease opening the door to earlier diagnoses and smarter therapies. Quantitative methods such as tandem mass tag (TMT), stable isotope labelling by amino acids in cell culture (SILAC) or label-free quantification (LFQ) approaches don’t just tell us which proteins are present, they also show us how much and under what conditions, unlocking entirely new insights for personalized medicine.
Proteomics for Tomorrow’s Medicine
Proteomics is just at the beginning of a new era with technologies that will fundamentally change our understanding of medicine.
AI-based protein design opens new ways to predict, model and even create entirely new proteins revolutionizing biotech and drug development.

Proteomics is undergoing a revolution. What once required years of work can now be done in hours: today's advanced mass spectrometry technologies allow us to analyze thousands of proteins at once with remarkable speed, precision, and from the smallest sample volumes. New technologies such as DIA (Data-Independent Acquisition) or single-cell proteomics even make it possible to record the activity of individual cells in detail. Powerful AI-driven analytics turn vast datasets into meaningful patterns that help distinguish health from disease opening the door to earlier diagnoses and smarter therapies. Quantitative methods such as tandem mass tag (TMT), stable isotope labelling by amino acids in cell culture (SILAC) or label-free quantification (LFQ) approaches don’t just tell us which proteins are present, they also show us how much and under what conditions, unlocking entirely new insights for personalized medicine.
Proteomics for Tomorrow’s Medicine
Proteomics is just at the beginning of a new era with technologies that will fundamentally change our understanding of medicine.
AI-based protein design opens new ways to predict, model and even create entirely new proteins revolutionizing biotech and drug development.
Approximately two thirds of our body weight is made up of fluids. Although blood accounts for only about 8% of this total, roughly 4.5 to 6 litres, it plays an essential role in maintaining our health. Blood transports oxygen and nutrients throughout the body, removes waste products, and supports both immune defense and wound healing. Far from being a uniform liquid, blood is a highly complex system composed of both cellular and fluid components. Its composition can vary slightly depending on factors such as age, overall health, immune status, and the body’s current fluid and electrolyte balance.
Blood cells - Specialists with Vital Roles
Roughly 45% of the blood volume consists of blood cells, each with distinct and critical functions:
Blood plasma - the Liquid Base
The remaining 55% of blood is composed of blood plasma, a clear, yellowish fluid that consists of around 90% water. The plasma also contains:
When clotting factors such as fibrinogen and prothrombin are removed from plasma, the resulting fluid is known as blood serum, commonly used in medical diagnostics.
Blood is more than a circulating fluid, it is a living mirror of human health. Flowing through every organ and tissue, it captures real-time snapshots of the body’s physiological and pathological states. As the most vital sample type in clinical diagnostics, blood has already led to the discovery of essential biomarkers, such as cardiac troponins for heart injury, insulin for diabetes, and C-reactive protein for inflammation. Yet, despite its vast potential, much of its proteomic landscape remains unexplored.
Mass spectrometry has revolutionized our ability to decode this complex matrix. With unmatched sensitivity, specificity, and scalability, it enables the simultaneous detection of thousands of proteins including novel biomarkers and isoforms that could redefine diagnostics.
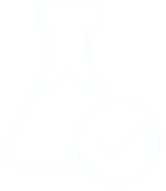
Plasma is one of the most information-rich yet analytically complex biological fluids due to its extraordinary dynamic range of protein concentration. Highly abundant proteins like albumin and immunoglobulins dominate the proteome, accounting for nearly 90% of the total protein mass, while clinically relevant low-abundance proteins may be present at concentrations up to 12 orders of magnitude lower. To overcome this challenge, pivotal innovation was introduced: the selective depletion of high-abundance proteins. Established several years ago, this technique allows researchers to unmask the rare and subtle signals hidden within the plasma proteome. By reducing the dominance of major proteins, mass spectrometry can now access a far broader spectrum of biologically and clinically significant molecules opening new frontiers in biomarker discovery, disease monitoring, and personalized medicine.
Beyond depletion, protein enrichment is a key step in unlocking the deeper layers of the plasma proteome. By selectively isolating specific protein groups such as low-abundance biomarkers, glycoproteins, or signaling molecules enrichment strategies allow us to focus on what truly matters: the biologically and clinically relevant signals often buried beneath the surface.
These techniques enhance the sensitivity and precision of mass spectrometry, enabling the detection of proteins that would otherwise go unnoticed. Enrichment opens new possibilities in biomarker discovery, disease profiling, and personalized medicine.

Beyond depletion, protein enrichment is a key step in unlocking the deeper layers of the plasma proteome. By selectively isolating specific protein groups such as low-abundance biomarkers, glycoproteins, or signaling molecules enrichment strategies allow us to focus on what truly matters: the biologically and clinically relevant signals often buried beneath the surface.
These techniques enhance the sensitivity and precision of mass spectrometry, enabling the detection of proteins that would otherwise go unnoticed. Enrichment opens new possibilities in biomarker discovery, disease profiling, and personalized medicine.
The power behind ProtiPrep’s™ 10-minute workflow lies in the seamless integration of two core innovations:
It is the synergy between our hardware and our chemistry that unlocks this unparalleled speed and simplicity. Together, they turn hours of complex, error-prone work into a single, efficient step. Explore below to learn how each component contributes to redefining proteomics sample preparation.
The ProtiPrep™ device is more than just a tube, it is a self-contained system engineered to eliminate complex handling and reduce pipetting errors. Its innovative two-part design consists of the upper Depletion Void (DV), which houses our proprietary resin, and the lower Sample Collection Tube. Every element is designed for one purpose: to make your workflow effortless. The user just pipettes 10 µL of plasma into the Depletion Void. The magic happens next: sealing the device with the screw-cap activates a unique buffer-release mechanism, seamlessly mixing the buffer with your sample and the resin. A brief 5-minute incubation with shaking, followed by a standard centrifugation step, is all it takes. After centrifugation, the device ensures a clean and easy separation. The user simply unscrews the top Depletion Void which traps the resin and all the bound, high-abundance proteins to reveal the Collection Tube. Inside is approximately 150 µL of purified, depleted plasma, ready for immediate downstream analysis. No tedious transfer steps, no risk of resin carry-over, and no sample loss, just a perfect sample, every time.

At the heart of every ProtiPrep™ device is our high-performance affinity resin, the engine driving our 10-minute workflow. We’ve optimized this chemistry for one purpose: to deliver maximum binding efficiency and reliability in record time.
Unmatched speed: The resin’s exceptionally rapid binding kinetics allow for the quick capture of high-abundance proteins. This is the key to achieving complete depletion in a protocol that takes just five minutes.
Powerful capacity: This speed is matched by a high dynamic binding capacity. Our resin is engineered to effectively remove even the most concentrated proteins, like albumin, without any risk of saturation.
Complete specificity: The powerful capture mechanism is balanced with optimized specificity. It targets abundant proteins while minimizing non-specific binding, ensuring your valuable, low-abundance biomarkers are protected and retained for analysis.
Working in perfect synergy with the ProtiPrep™ device, our resin is your guarantee for clean, consistent results in a fraction of the time.